By Nelson Vergel
Author of “Testosterone: A Man’s Guide”
There is so much confusion about the use of hormones in pre and post menopausal women, especially after the largest study ever undertaken using two female hormones was halted due to increased incidence of breast cancer in women. I will try to attempt to distill issues related not only to female based hormone therapy (estrogen and progestin) but also to the use of testosterone replacement therapy in women.
WHAT IS TESTOSTERONE? ISN’T IT A MALE HORMONE?
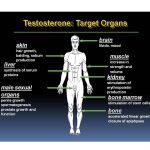
Testosterone is the hormone responsible for normal growth and development and maintenance of male sex characteristics. It also affects lean body mass, mood and sexual function in both males and females, so it is not only a “male” hormone. It is the primary androgenic (responsible for masculine characteristics) and anabolic (muscle building) hormone.
Testosterone is produced by the testicles in males and by the ovaries in females, with small amounts also produced by the adrenal glands in both genders. Men produce about 7-8 times more testosterone than women.
Testosterone is also the precursor of estradiol (a “female” hormone) in men and women.
SEXUAL DYSFUNCTION IN WOMEN
As we all know if we watch TV for more than an hour, we are bombarded daily by ads selling erectile dysfunction drugs for men. But we hardly hear anything about sexual dysfunction in women, and there are many reasons why women’s sexual desire and other hormone related quality of life issues are so misunderstood, under-diagnosed and under treated.
There is no FDA approved product for the treatment of sexual dysfunction in women. There is also no FDA approved testosterone replacement product for women in 2012, although some physicians are prescribing it to women in an “off-label” manner (legal but not for the FDA approved “intended use”) using low doses of commercial products approved for men (Androgel, Testim, Fortesta, Testopel, Depo Testosterone and Axiron) or via compounded pharmacy gel/cream formulations.
Sexual dysfunction in pre and post-menopausal women has been a very controversial topic that has been poorly researched even though a February, 1999 study published in the Journal of the American Medical Association, titled, “Sexual Dysfunction in the United States: Prevalence and Predictors,” found that approximately 43% of postmenopausal women suffer from some form of female sexual dysfunction.
It wasn’t until June 2011 that an FDA advisory committee to the division of Reproductive and Urologic Drug Products stated that HSDD (Hypoactive Sexual Desire Syndrome) is a significant medical condition for women. This may open the door for companies to apply for new drug applications for that indication.
It is important to note that unlike erectile dysfunction drugs approved for men like Viagra that increase blood flow to the genitals as long as a man is aroused, testosterone therapy is systemic and needs to be applied over a period of weeks to have a noticeable effect on sex drive in men and women.
Some companies have tried to enter the female sexual dysfunction market in the past. In December 2004 the United States theFDA rejected Procter and Gamble’s fast-track request for Intrinsa (a testosterone patch for women) for HSDD citing concerns about potential off-label use of the product was to be approved. In Canada, post-menopausal women have been able to obtain government-approved testosterone treatment since 2002. In 2007, Intrinsa was granted a license from the European Medicines Agency in July, and is available on Britain’s National Health Service.
According to a P&G’s survey on female health, 30 million women in the U.S. are naturally menopausal, 3 million are distressed by their lack of sexual desire, and 20% of 25 million women who are surgically menopausal are distressed.
Other companies that attempted to get their drugs approved for this indication (Boehringer Ingelheim and Warner Chilcott) have each pulled the plug on their competing HSDD treatments for menopausal and pre-menopausal women.
BioSante Pharmaceuticals is the only company left in the race for a treatment for HSDD. This company is in late stage clinical studies using LibiGel (testosterone gel) treatment of HSDD but has had issues in the approval of this product by the FDA. More on this product at the end of this article.
HORMONE THERAPY IN POST MENOUPAUSAL WOMEN
Menopause can cause symptoms such as hot flashes might result from the changing hormone levels during the menopause transition. After a woman’s last menstrual period, when her ovaries make much less estrogen and progesterone, some symptoms of menopause might disappear, but others may continue.
To help relieve these symptoms, some women use hormones. This is called hormone therapy (HT) which includes estrogen alone or in combination with progestin. HT is available orally or in gel formulations made by specialized compounding pharmacies.
Estrogen is a hormone used to relieve the symptoms of menopause. Estrogen alone (E) may be used by a woman whose uterus has been removed. But a woman who still has a uterus must add progesterone or a progestin (synthetic progesterone) along with the estrogen (E+P). This combination lowers the chance of an unwanted thickening of the lining of the uterus and reduces the risk of cancer of the uterus, an uncommon, but possible result of using estrogen alone.
But the use of hormone therapy in women has been subject of a lot on controversy in recent past.
Researchers from the Harvard School of Public Health analyzed data from the landmark Women’s Health Initiative (WHI) clinical trial of the effects of combination hormone therapy (estrogen+ progestin) in 16,608 postmenopausal women with an intact uterus, ages 50 to 79 years (average age of 63) at enrollment. This study did not include the use of testosterone. In this study, 8,506 participants were randomly assigned to receive a combination of estrogen (0.625 milligrams of conjugated equine estrogens per day) plus progestin (2.5 mg of medroxyprogesterone acetate), and 8,102 women were given placebo.
The study was stopped in 2002 after an average of 5.6 years of treatment due to an increase in breast cancer in the women on hormone therapy. Compared to women on placebo, women on combination hormone therapy were also at increased risk of stroke, dangerous blood clots, and heart disease, while their risk of colorectal cancer and hip fractures was lower.
It is important to note that these hormones were provided orally and that some clinicians claim that transdermal (on the skin) application of estrogen alone or in combination with testosterone would show a different and more favorable side effect profile.
The halting of the WHI study raised concerns about safety of all hormone therapy in women (oral or transdermal), even if no androgens will included in this study and only oral delivery forms were used. Many physicians stopped prescribing hormone therapy (HT) even for women who had dramatic improvements in their quality of life while using it. Experts today don’t recommend hormone therapy unless a woman suffers difficult menopause symptoms.
Emerging data on the use of androgens (testosterone and DHEA) alone or in combination with HT are showing that there may be potential benefits of these hormones in women with androgen deficiency and sexual dysfunction.
Androgens are also precursors of all estrogens (estrone (E1), estradiol (E2), and estriol (E3)) in women’s bodies. The primary and most well-known androgen is testosterone (which aromatizes into estradiol), other less important androgens are dihydrotestosterone (DHEA) and androstenedione. Androgens are directly secreted by the ovaries and adrenals in women.
Presently, there is no agreement about whether androgen deficiency is a clinical problem in aging women and if the addition of androgens to HT can ameliorate the cardiovascular risks seen in the WHI study.
Causes of androgen insufficiency in women can have ovarian, adrenal, hypothalamic-pituitary, drug-related, and unknown origins. Symptoms of androgen insufficiency in women may include a diminished sense of well-being, low mood, fatigue, and hypoactive sexual desire disorder (HSDD) with decreased libido, or decreased sexual receptivity and pleasure that causes a great deal of personal distress.
There is increasing evidence to suggest that many postmenopausal women experience symptoms alleviated by androgen therapy and that such symptoms may be caused by androgen deficiency. Affected women complain of fatigue, low libido, and diminished well-being, which are symptoms easily and frequently attributed to psychosocial and environmental factors.
The question of whether adding testosterone therapy to conventional postmenopausal HT is effective or safe is unresolved. Therefore, a Cochrane review was performed to determine the efficacy and safety of testosterone therapy for postmenopausal women using HT. Thirty-five trials with a total of 4768 participants were included in the review. The median study duration was six months (range 1.5 to 24 months). Most of the trials were of adequate quality with regard to randomization. The pooled estimate suggested that the addition of testosterone to HT regimens improved sexual function scores and number of satisfying sexual episodes for postmenopausal women. Some of the few adverse effects were decreased high-density lipoprotein (HDL) cholesterol levels and an increased incidence of hair growth and acne. The discontinuation rate was not significantly greater with the addition of testosterone therapy.
OTHER USES IN WOMEN
Emerging and controversial potential indications for androgen therapy in women have been or are being evaluated. These include use in women with premature ovarian failure, premenopausal androgen deficiency symptoms, postmenopausal and glucocorticosteroid-related bone loss, treatment of HIV related wasting, and management of premenstrual syndrome. Whether or not any of these indications will actually have approved products in the future is unknown.
HYPOGONADISM IN MEN, BUT WHAT TO CALL IT IN WOMEN?
The term hypogonadism is used as a diagnosis term for testosterone deficiency in men. Besides HSDD as one of the potential symptoms, there is no agreement on what to call androgen deficiency in women. In clinical guidelines published in 2002 called the Princeton consensus statement (Fertil Steril. 2002 Apr;77(4):660-5) used the term “female androgen insufficiency” as defined as by a pattern of clinical symptoms in the presence of decreased bioavailable testosterone and normal estrogen status .
The panel warned that currently available testosterone assays were found to be lacking in sensitivity and reliability at the lower testosterone blood level ranges present in women, and the need for an equilibrium dialysis measure was strongly emphasized as the most adequate method to test women’s testosterone blood levels.
TESTOSTERONE REPLACEMENT IN WOMEN- POTENTIAL BENEFITS
Circulating testosterone in women declines during the late reproductive years such that otherwise healthy women in their 40s have approximately half the testosterone level as women in their 20s. The levels remain stable across the menopausal transition and then either remain stable or continue to decline with diminishing adrenal androgen production with increasing age. In the decade preceding menopause, there is loss of the mid-cycle surge of free testosterone. Despite this, research showing the benefits of androgen replacement has been limited to the postmenopausal years.
Some small studies have been done in premenopausal women, however. One evaluated the efficacy of transdermal testosterone therapy on mood, well-being, and sexual function in eugonadal (normal testosterone blood levels), premenopausal women presenting with low libido. Testosterone therapy improved well-being, mood, and sexual function in these women. Since a substantial number of women experience diminished sexual interest and well-being during their late reproductive years, further research is warranted to evaluate the benefits and safety of longer-term intervention. Potential side effects that are dose dependent may be unwanted hair growth, masculinization, and lowering of high density lipoprotein (HDL).
TESTOSTERONE IN HIV+ WOMEN:
Dr Bhasin and his team (JCEM 83 (4): 1312) has found that the higher the HIV viral load, the lower the blood levels of testosterone in HIV+ women. In general, HIV+ women had lower testosterone than HIV- women. Low testosterone was not correlated to unintentional weight loss in Bhasin’s data.
In healthy women, the ovaries and the adrenal glands contribute equally to the maintenance of circulating testosterone levels. Bhasin presumes that there may be some HIV effect on the ovaries that may be one of the root causes of testosterone deficiency in women with HIV.
In HIV+ women with unintentional weight loss, a placebo controlled study using testosterone a testosterone patch did not improve lean mass and strength, although Bhasin mentions that the study was not powered to determine if women with low serum free testosterone at baseline had different gains that those who had normal testosterone (the study allowed any baseline testosterone blood level as long as there had been a weight loss of 5% or more at baseline).
A study done by Dr Grinspoon’s team (Arch Intern Med. 2004 Apr 26;164(8):897-904.) showed that low dose testosterone supplementation in HIV+ women with unintentional weight loss increased lean body mass and strength. The study did not specify how many of these women had low testosterone at baseline, however.
Presented in CROI-2009 (Abstract 976), a 18 month randomized placebo-controlled study performed by Dr. Labby and colleagues from Harvard Medical School investigated the effects of transdermal (patch) testosterone replacement (300 mcg twice weekly vs. placebo) on body composition, bone density, metabolic parameters, quality of life, and safety among relatively androgen deficient women with HIV.
Significantly benefits were seen with the use of testosterone replacement in body composition, quality of life and bone density in HIV positive women who had baseline free testosterone levels below 3 picograms per milliliter (pg/mL) (the normal range is between 1.1 and 6.3 pg/m). Testosterone use was well tolerated without virilizing effects and did not affect lipids, liver function, or HIV viral load.
Most HIV+ women that I talk to have never had their testosterone blood levels checked. Unfortunately, most HIV clinical practices in the United States have not recognized androgen deficiency diagnosis and treatment even in well insured women with symptoms of low sex drive, fatigue, loss of lean body mass, and menstrual irregularities.
TESTOSTERONE IN HIV+ WOMEN WITH LIPODYSTROPHY
Dr Grispoon and his team have surprisingly found that free serum testosterone was three times higher in women with lipodystrophy compared to those without lipodystrophy and healthy controls (J Clin Endocrinol Metab. 2000 Oct;85(10):3544-50). In the past 15 years lecturing on metabolic disorders in HIV around the country, I have met dozens of HIV+ women that have been on antiretrovirals for years who have some signs of masculinization (facial hair growth and wider jaw line). Some had been diagnosed with Polycystic Ovary Syndrome (PCOS) but others had never been examined for this diagnosis.
Women with Polycystic Ovary Syndrome (PCOS) have higher levels of testosterone and insulin in general. The main symptoms of PCOS are menstrual irregularities, hirsutism, acne, infertility metabolic syndrome, and others. Metformin, an insulin sensitizer, and finasteride, a dehydrotestosterone (DHT) inhibitor, have been shown to normalize testosterone in women with PCOS.
Dr Grispoon and his team found that HIV+ women with severe lipodystrophy had no higher incidence of PCOS, but some other reports contradict this finding (JCEM 90 (10): 5596).
So, is higher testosterone in HIV+ women with lipodystrophy caused by HIV, inflammatory factors, or multifactorial? I do not think we will ever find out, but I think it is prudent to measure total and free testosterone in HIV+ women with lipodystrophy that may be experiencing menstrual problems and/or excessive hair growth.
TESTOSTERONE REPLACEMENT AND CARDIOVASCULAR RISKS IN WOMEN
A direct association between testosterone and heart disease has never been established, but for many years, doctors have suspected that a link exists. The reasoning goes like this: men have much more testosterone than women, and they develop heart disease about 10 years before their female counterparts.
Women with systolic heart failure who took low-dose testosterone for six months, on top of standard medical therapy, showed significant gains in exercise and ventilatory capacity and large-muscle strength along with heightened insulin sensitivity, in a small placebo-controlled trial ( J Am Coll Cardiol 2010; 56:1310-1316).
Despite the entrenched belief that higher blood levels of testosterone increase the risk of CVD in women, data from recent observational studies mostly show an inverse relationship between testosterone and CVD risk. A pilot study (JCEM 86 (1): 158) suggested favorable effects of transdermal testosterone treatment of women with established congestive cardiac failure which merits further evaluation. Preliminary data indicate that injectable testosterone therapy improves both endothelial-dependent (flow-mediated) and endothelium-independent (GTN-mediated) brachial artery vasodilation in postmenopausal women using long-term estrogen therapy. The mechanisms underlying these potentially beneficial cardiovascular effects require further investigation.
TESTOSTERONE REPLACEMENT AND CANCER IN WOMEN
The relationship between endogenous testosterone production and breast cancer risk remains contentious, with recent studies indicating either no relationship, or a possible increase in risk when estrone and estradiol are not taken into account. No randomized controlled trial of testosterone therapy has been sufficiently large or of sufficient duration to establish whether such treatment may influence breast cancer occurrence. There does not appear to be an association between testosterone and endometrial cancer, or other malignancies on review of published studies.
There is now convincing evidence that usual estrogen based hormone therapy for ovarian failure increases the risk for breast cancer. However, some studies have previously shown that ovarian androgens normally protect mammary epithelial cells from excessive estrogenic stimulation, and therefore a study hypothesized that the addition of testosterone to usual hormone therapy might protect women from breast cancer (Menopause: Sept/Oct 2004 – Volume 11 – Issue 5 – pp 531-535). This was a retrospective, observational study that followed 508 postmenopausal women receiving testosterone in addition to usual hormone therapy in South Australia. Breast cancer status was ascertained by mammography at the initiation of testosterone treatment and biannually thereafter. The average age at the start of follow-up was 56.4 years, and the mean duration of follow-up was 5.8 years. Breast cancer incidence in this group was compared with that of untreated women and women using usual hormone therapy reported in the medical literature and to age-specific local population rates.
There were seven cases of invasive breast cancer in this population of testosterone users, for an incidence of 238 per 100,000 woman-years. The rate for estrogen/progestin and testosterone users was 293 per 100,000 woman-years-substantially less than women receiving estrogen/pro-gestin in the Women’s Health Initiative study (380 per 100,000 woman-years) or in the Million Women Study (521 per 100,000 woman-years). The breast cancer rate in testosterone users in this study was closest to that reported for users who never used hormone therapy in the latter study (283 per 100,000 woman-years), and their age-standardized rate was the same as for the general population in South Australia.
These observations suggest that the addition of testosterone to conventional hormone therapy for postmenopausal women does not increase and may indeed reduce the hormone therapy-associated breast cancer risk-thereby returning the incidence to the normal rates observed in the general, untreated population. But more studies are needed.
CURRENT USE IN THE UNITED STATES
As previously mentioned, there is not a FDA approved testosterone product for women. The few physicians that are prescribing it are doing so by having their female patients use low doses of products approved for male hypogonadism (Androgel ,Testim, Axiron, Testopel and Fortesta) in an off label manner, or prescribing gels with small testosterone concentrations via compounding pharmacies.
LIBIGEL, THE FIRST GEL THAT COULD HAVE BEEN APPROVED IN THE UNITED STATES FOR WOMEN
On January 24, 2008, the US FDA notified BioSante that it had completed and reached agreement with BioSante on a Special Protocol Assessment (SPA) for BioSante’s Phase III safety and efficacy clinical trials of LibiGel in the treatment of hypoactive sexual desire syndrome (HSDD).
Both Phase III safety and efficacy trials were finished and were double-blind, placebo-controlled trials that have enrolled approximately 500 surgically menopausal women each for six-months of treatment.
Unfortunately, BioSante Pharmaceuticals reported in December of 2011 that LibiGel did not meet the co-primary or secondary endpoints in two pivotal Phase III efficacy trials in the treatment of hypoactive sexual desire disorder in postmenopausal women, so the FDA did not approve it.
The co-primary endpoints of both LibiGel efficacy trials were the change in the total number of days with a satisfying sexual event from baseline, and the change in mean sexual desire from baseline. The secondary endpoint for both trials was the change in sexual distress from baseline.
Women in the first trial, called BLOOM-1, who were treated with LibiGel showed an increase of 1.47 days with a satisfying sexual event compared to baseline, while those receiving placebo gel showed an increase of 1.26 days with a satisfying sexual event compared to baseline.
Women in BLOOM-2 who were treated with LibiGel showed an increase of 1.0 day with a satisfying sexual event compared to baseline, while those receiving placebo gel showed an increase of 1.28 days with a satisfying sexual event compared to baseline.
Although there were no statistical differences in the endpoints, BioSante said all results were in the appropriate directions. The LibiGel groups in both trials showed an increase in free testosterone levels compared to baseline and placebo.
BioSante continues to conduct the Phase III LibiGel safety study, a cardiovascular events and breast cancer study that has completed enrollment of 3,656 women and has accrued over 5,100 women-years of exposure, to date. The study is designed for a total of five years; however, BioSante could use the safety study data as part of a New Drug Application (NDA) submission after the last subject enrolled has completed 12 months of exposure to LibiGel or placebo.
CONCLUSION
In conclusion, testosterone replacement in androgen deficient women with or without HIV may have a good risk-to-benefit ratio for some patients, but conflicting data, lack of long term studies and lack of a FDA approved product for that indication may be barriers to making it part of standard of care of aging women. The approval of a testosterone product specifically for women in the future may change that.
More studies need to be performed with validated and meaningful endpoints. In the meanwhile, many women are not waiting for long term studies and instead are getting off-label prescriptions of compounded testosterone from physicians who specialize in female hormone balance.